Neural circuits that help us find our way home - and how they are impaired in neurological & psychiatric disorders
Charles Darwin used the term “dead reckoning” to describe the mysterious ability of most species to seamlessly navigate back home, even in the absence of external guiding cues. Finding our way home is indeed something most of us take for granted, but this facility is dramatically impaired in many people with Alzheimer’s disease, Parkinson’s disease, and epilepsy. Our research is motivated by a desire to understand how healthy neurons in the brain compute the way home and how these same neuronal computations fail in neurological and psychiatric disorders, with the ultimate goal of therapeutically repairing these neurons and computations.
Rational rules for the use of psychedelic medicine, including the custom engineering of multiple new mouse lines
Psychedelic medicine makes many assumptions about what neurons and brain regions are tarteged by psychedelic drugs, as well as how neurons are supposedly altered. We work to causally validate or debunk these assumptions using systematic physiology, pharmacology, AI-driven data assimilation, and behavioral analyses. A rigorous experimental understanding together with computational rigor is necessary to identify the utility and limits of psychedelic medicine. To enable this, we have also engineered multiple new mouse lines and look forward to sharing these so that we can collaboratively decipher how psychedelic drugs impact healthy and neuropsychiatric circuits.
Read on for our papers, and please get in touch with Omar if you have questions about our research, want to support our work, or if you’re interested in joining or collaborating with the lab.
New Manuscripts from the Lab:
psychedelic therapeutic utility IS BROADER than currently appreciated
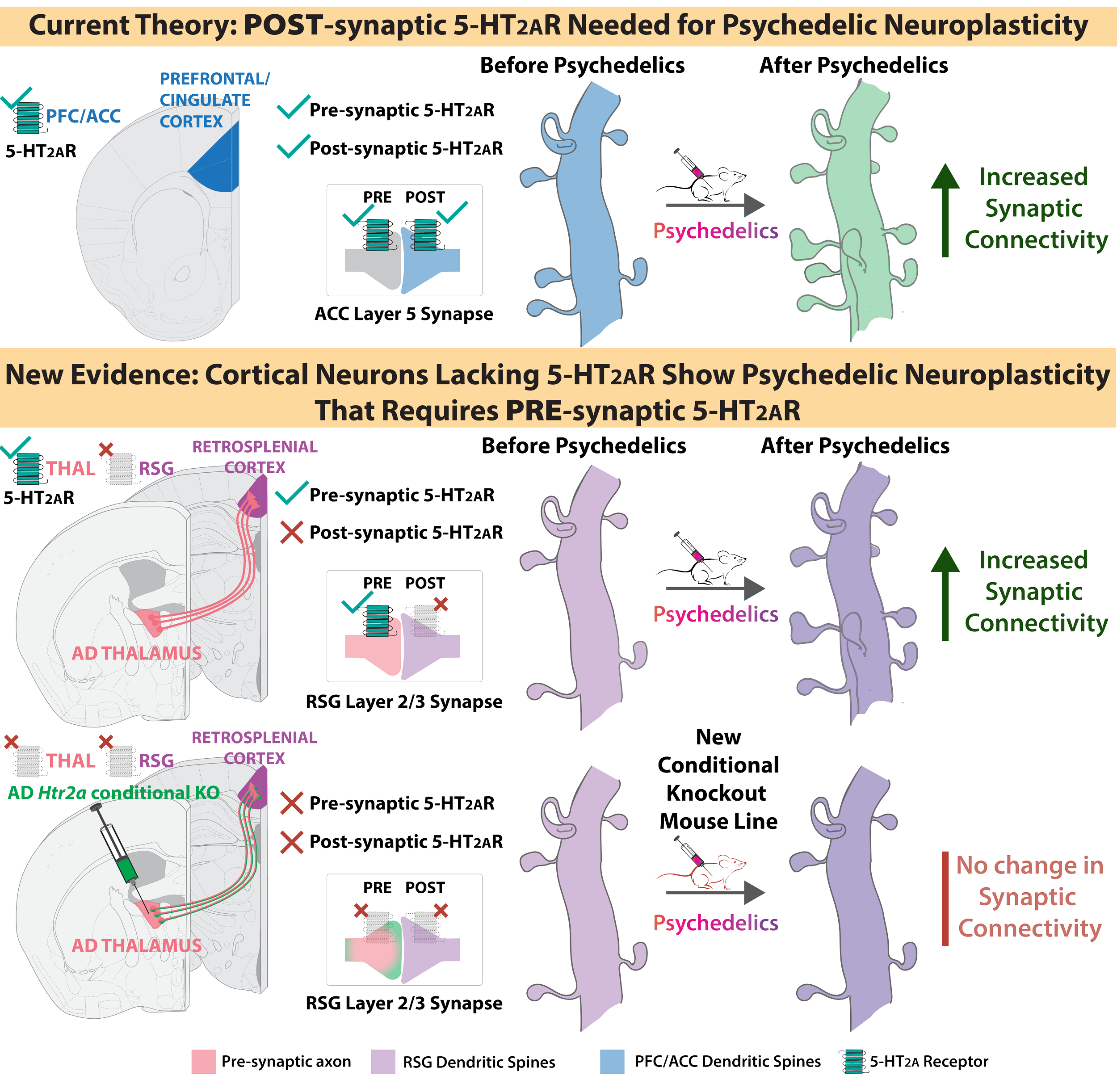
Ekins TG*, Rybicki-Kler C*, Deng T, Brooks I, Jedrasiak-Cape I, Donoho E, Ahmed OJ (2025). Psychedelic neuroplasticity of cortical neurons lacking 5-HT2A receptors. Molecular Psychiatry
The Defining Cell Type of the Granular Retrosplenial Cortex is Radically Different
Jedrasiak-Cape I, Rybicki-Kler C, Brooks I, Ghosh M, Brennan EWK, Kailasa S, Ekins TG, Rupp A, Ahmed OJ (2025). Cell-type-specific cholinergic control of granular retrosplenial cortex with implications for angular velocity coding across brain states. Progress in Neurobiology, 251:102804
PDF HTML PRESS: U-M scientists discover unique brain cell that may hold key to Alzheimer’s disorientation
Weeks-long boost in cognition with single-dose psychedelic
Brouns EJ, Ekins TG, Ahmed OJ (2025). Single-dose psychedelic enhances cognitive flexibility and reversal learning in mice weeks after administration.
PAPER PRESS: DISCOVER MAGAZINE UM NEWS INSIDE PRECISION MEDICINE ALTMETRIC: Top 5% of all research output
A definitive transcriptomic atlas of retrosplenial neurons across species
Psychedelics Decrease Excitability & Working Memory in Prefrontal Cortex
Ekins TG, Brooks I, Kailasa S, Rybicki-Kler C, Jedrasiak-Cape I, Donoho E, Mashour GA, Rech J, Ahmed OJ (2023). Cellular rules underlying psychedelic control of prefrontal pyramidal neurons.
Left-Right Brain Communicate Better When Running Faster or in REM Sleep
Ghosh M, Yang FC, Rice SP, Hetrick V, Lorenzo A, Siu D, Brennan EW, John TT, Ahrens AM, Ahmed OJ (2022). Running speed and REM sleep control two distinct modes of rapid interhemispheric communication. Cell Reports 40:111028
PDF HTML PRESS: PSYCHOLOGY TODAY UM PRESS RELEASE
Parallel Processing in Superficial Retrosplenial Ctx
Brennan EW*, Jedrasiak-Cape I*, Kailasa S, Rice SP, Sudhakar SK, Ahmed OJ (2021). Thalamus and claustrum control parallel layer 1 circuits in retrosplenial cortex. eLife, 10:e62207
PDF HTML eLife DIGEST PRESS Chosen as BIOMEDICAL PICTURE OF THE DAY
Human Inhibitory Neurons Dynamically Control Seizures
Ahmed OJ, John TT, Sudhakar SK, Brennan EKW, Lorenzo Gonzalez A, et al. (2020). Two modes of inhibitory neuronal shutdown distinctly amplify seizures in humans. Submitted
Speed Distinctly Controls the Amplitude vs Shape of Theta
Ghosh M*, Shanahan BE*, Furtak SC, Mashour GA, Burwell RD, Ahmed OJ (2020). Instantaneous amplitude and shape of postrhinal theta oscillations differentially encode running speed. Behavioral Neuroscience 134:6 516–528